We primarily engaged in the development of novel and potential first-in-class drugs with the potential of becoming paradigm-shifting blockbuster therapies for cardiovascular diseases with large patient populations and significant innovation gaps. We have developed a robust pipeline with five drug candidates:
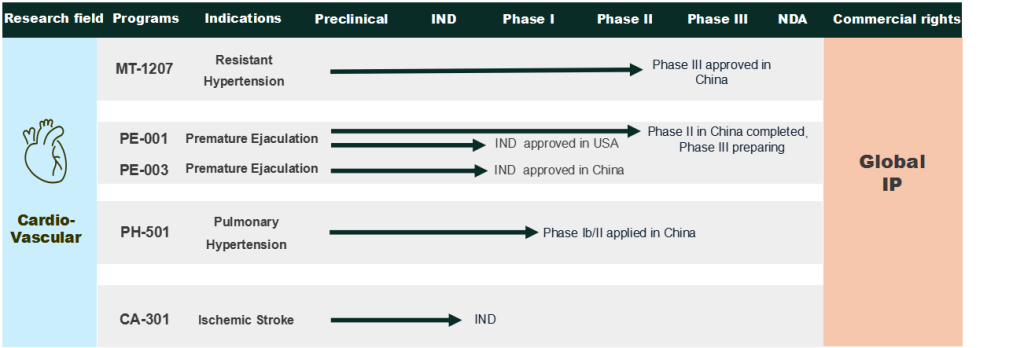
Pulmonary hypertension (PH) is a serious, life-threatening, and progressive disease affecting the structure or function of pulmonary blood vessels, which, in severe cases, may lead to right heart failure or even death, often referred to as the cancer of cardiovascular disease. Effective treatment options are very limited, with many patients experiencing low therapeutic benefits that will often decline over time. Our PH-501, featuring a novel mechanism of action and multi-target design, has shown promise in preclinical studies, with significant decreases observed in right ventricular hypertrophy index, right ventricle to body weight ratio, right ventricular free wall thickness, and right ventricular systolic pressure, each being key efficacy indicators in PH treatment. We are currently conducting a phase 1b/2 clinical trial.
Premature ejaculation(PE)is a severely underdiagnosed and undertreated men’s health disease, with only one approved new molecular entity PE drug — dapoxetine globally, and it is expected that there will be significant growth potential for PE drugs. Our PE-001 is the only NME drug candidate currently under clinical development in China. With a novel mechanism of action and multi-target design, our PE-001 is positioned as a first-in-class NME PE drug candidate and potentially the second NME drug approved for PE globally. We have currently completed the Phase II clinical trial and are actively preparing for Phase III.
Stroke is a leading cause of death and disability worldwide. The prevalence of ischemic stroke in China is significant and growing, expected to increase to 31.3 million in 2032 from 21.5 million in 2023. Current frontline ischemic stroke treatments face significant limitations, including a narrow therapeutic window, bleeding risks, and limited efficacy in large-vessel occlusions, signifying the vast demand for effective treatment. Our CA-301 demonstrated encouraging therapeutic activities in preclinical animal studies, significantly reducing cerebral infarct volume compared to the model control group and improving neurological function. We plan to file an IND application for CA-301 by the first quarter of 2026.
Hypertension is one of the most common chronic cardiovascular diseases, with over 346.5 million patients in China in 2023. Five categories of first-line treatments have been available for over a decade, but only 11% of adult hypertension patients in China have effectively controlled their blood pressure. More importantly, around 15% of patients suffer from resistant hypertension (RH), characterized by high blood pressure that does not respond adequately to intensive medical treatment. With a novel mechanism of action and triple-target design, our MT-1207 is positioned to disrupt the current hypertension treatment paradigm, with the potential to become the sixth and its own category of first-line treatment, particularly for resistant hypertension. We have currently completed the Phase II clinical trial and already got approved for Phase III.
